This Gummy Bear Experiment is a fun activity for children of a variety of ages. Though playing with food is usually not encouraged, this fun experiment is a great way for children to learn the basics of chemistry. This fun project that exploration and discovery is a great way for children to explores osmosis.
Check out the list of materials that we use with these printables.
Osmosis
Usually, most candy dissolves in water. The gelatine in gummy bears prevent them from dissolving. By using these sugary sweets in this experiment, children will be learning about osmosis.
But what is osmosis? It is the process of water moving from a greater concentration of water to a lower concentration of water.
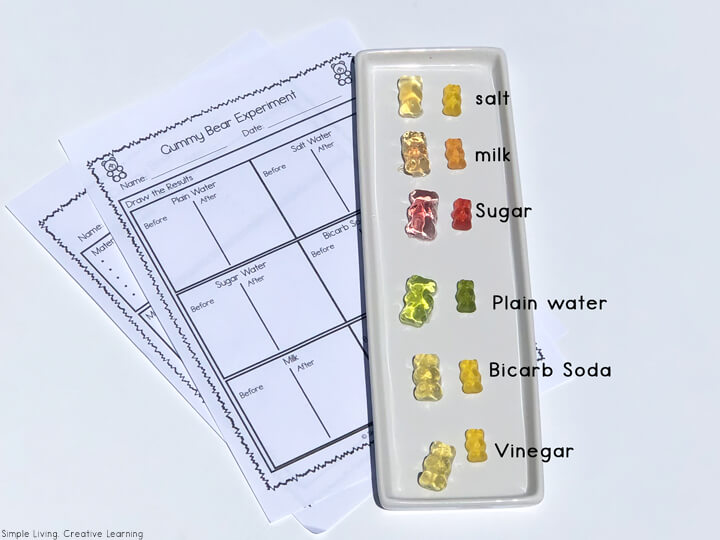
Gummy Bear Experiment Supplies
To complete this experiment, you will need a packet of Gummy Bears. I used these ones that I purchased from Woolworths (Australia). Then you will need to print off the experiment printables.
You will also need different liquids such as:
- water – room temperate and boiling
- milk
- vinegar
- salt
- sugar
- bicarbonate of soda (baking soda)
Let’s Grow Gummy Bears
Start by completing the parts of the experiment printables that you can, such as the Materials Needed, Hypothesis as well as the before pictures. We also measured the gummy bears and weighed them.
Then collect your cups and add the water, milk, vinegar or other fluids that you are using. In the picture below, you will see that the cups are quite full. We ended up removing half of the water, just after this photo was taken.
For young children, try using small plates that are more open for them to see what is happening better.
For the ingredients that are added to the water, such as salt, boil some water and slowly add a teaspoon of salt, mixing it as you go. Keep adding salt and mixing in.
You will end up with around 1 tablespoon of salt to ½ cup of boiling water. Let it cool completely, even place it in the fridge before adding your bear as you don’t want to melt it.
Repeat as above with the boiling water for the sugar and bicarb soda water, allowing to cool before starting.
Add one or two bears to each of the containers.
After about 18 hours, we checked our bears and recorded our data. The kids drew the results as well as measured the bears. If you leave them too long, they will start to break or fall apart.
Note
I suggest that you don’t eat them as not only would they taste horrible, they would probably be full of bacteria from sitting in the containers and with all the handling of them.
The Science Behind This
The main ingredients of these bears are sugar, sucrose and gelatine. The gelatine absorbs some of the water, allowing the bears to expand, without dissolving, which is a great way to teach kids about osmosis. What is osmosis? Since the bears do not contain water, the water moves into the bear by the process of osmosis – which is the process where water moves from a greater concentration of water to a lower concentration of water. Over time, the bear acts like a sponge, absorbing the water rather than being dissolved in it.
Alternative Experiments
This experiment doesn’t have to end there! It can be changed up, simply by
- increasing the amount of sugar, salt etc.
- decreasing the amount of sugar, salt etc.
- testing using different liquids
- weighing your gummy bears and create a graph or chart to demonstrate how it changes over a certain time period
Looking for more exciting Science Experiments? Why not try these?
- Strawberry DNA Experiment
- Pop Rock Experiment
- Rainy Day Experiment
- Walking on Eggs Experiment
- Marshmallow Stem Building Activity
For older children, have them fill out these Scientific Method Flip Book and Worksheets when completing this experiment.





 Make your Own Watercolour Paints
Make your Own Watercolour Paints